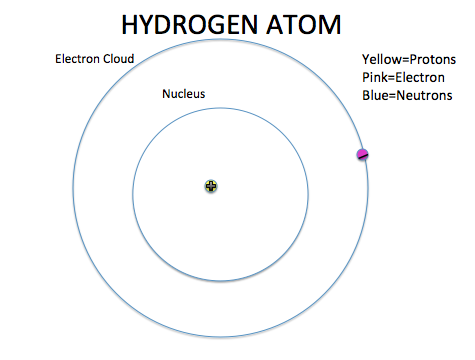
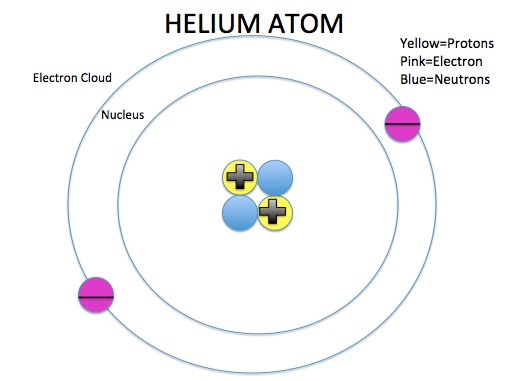
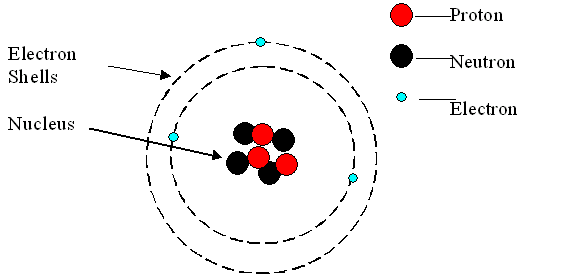
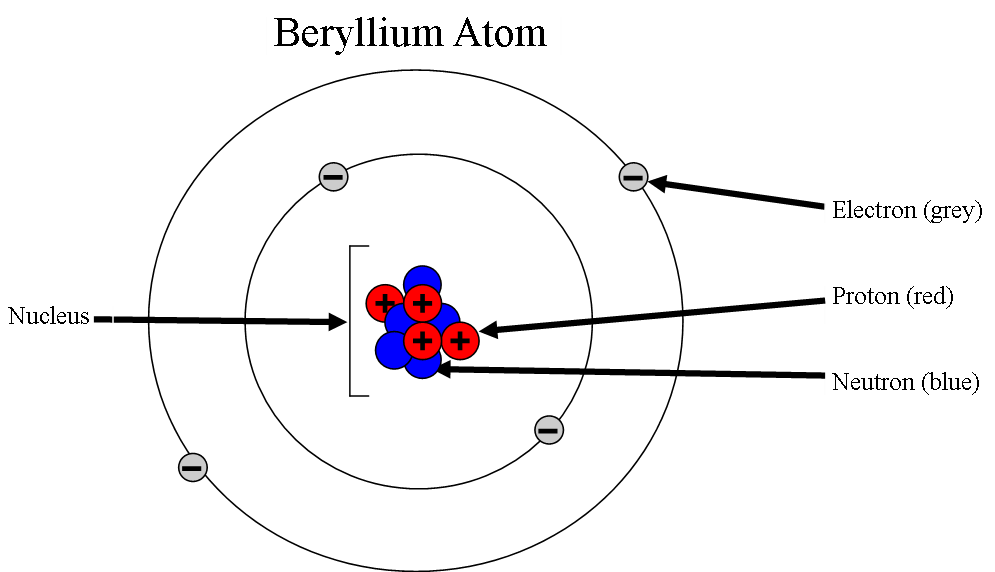
- The mass of an atom is found in the nucleus
- The electron cloud contributes the most to the volume of an atom
- The nucleus has a positive charge because of the protons ALWAYS
- The electron cloud has a negative charge because of the electrons ALWAYS
- Opposite forces attract so the atom stays together
- PARTICLES DETERMINE THE ATOM
Lithium Atom
1. Electrons have a negative charge and are found in the are of an atom called the electron cloud.
2. The atomic number of an atom is also the number of protons or electrons in an atom.
3.Protons have a positive charge and are found in the area of an atom called the nucleus.
4.Neutrons do not have a charge and are found in the area of an atom called the nucleus
5. Protons and neutrons contribute mass to the atom.
6. Electrons contribute to an atom's volume.
7. Each proton and each neutron contribute 1 amu to an atom. And amu means atomic mass unit
8. If we know the total mass of atom in amu, we can subtract the number of protons to calculate the number of neutrons found in the nucleus of the atom.
9. In a neutral atom, the number of protons is the same as the number of electrons.
10. Atomic numbers for atoms increase in increments of one for the elements on the periodic table of elements.
11. An atom is made up of a nucleus and an electron cloud. Electron clouds are where electrons are found. The nucleus is where the protons and neutrons are found.
12. The number of protons are what define an atom (different subatomic particles).
2. The atomic number of an atom is also the number of protons or electrons in an atom.
3.Protons have a positive charge and are found in the area of an atom called the nucleus.
4.Neutrons do not have a charge and are found in the area of an atom called the nucleus
5. Protons and neutrons contribute mass to the atom.
6. Electrons contribute to an atom's volume.
7. Each proton and each neutron contribute 1 amu to an atom. And amu means atomic mass unit
8. If we know the total mass of atom in amu, we can subtract the number of protons to calculate the number of neutrons found in the nucleus of the atom.
9. In a neutral atom, the number of protons is the same as the number of electrons.
10. Atomic numbers for atoms increase in increments of one for the elements on the periodic table of elements.
11. An atom is made up of a nucleus and an electron cloud. Electron clouds are where electrons are found. The nucleus is where the protons and neutrons are found.
12. The number of protons are what define an atom (different subatomic particles).