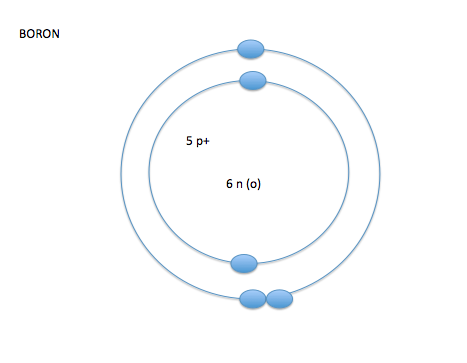
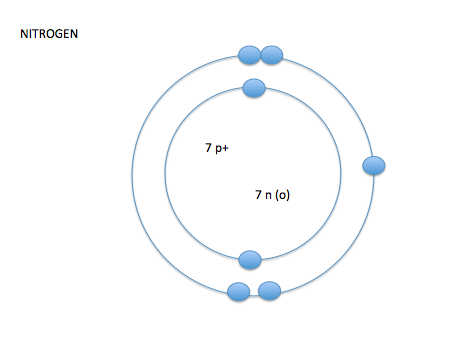
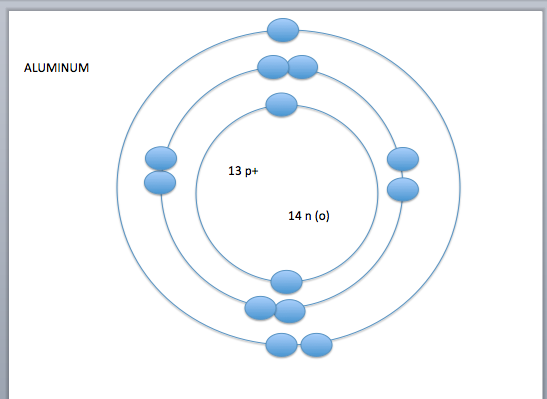
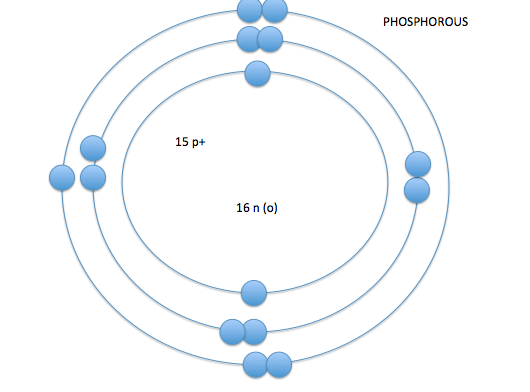
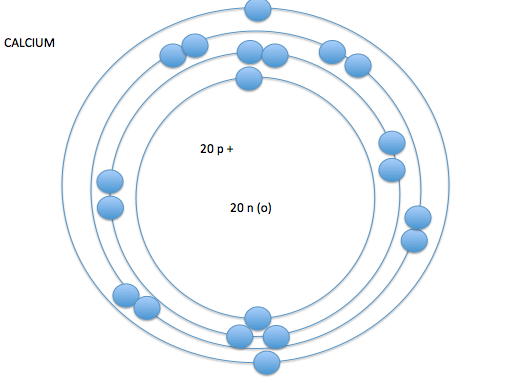
- the electron cloud is broken into seven layers
- these layers of the electron cloud are called energy levels or energy shells
- on energy level 1 you can have up to 2 electrons, on energy levels 2-7 you can have up to 8 electrons*
- the number of electrons in the outer layer control how or if atoms react with other atoms- valence electrons
- valence shell-the outermost shell of an atom
- valence electrons- amount of electrons on the outermost shell
OCTET RULE- atoms tend to want their outside energy level full of electrons
- If a neutral atom's valence shell is full, then the atom will not react with other atoms.
- If a neutral atom's valence shell has space available, then the atom will react with other atoms based on the number of electrons found in the valence shell
some atoms tend to lose electrons and become a positive ion
some atoms share electrons and that does not affect charge
VALENCE DROP, ADD, OR SHARE?
1. Helium (exception), argon, and neon all are balanced atoms.
2. No because balanced electrons won't gain or lose electrons
3. They have 4 valence electrons
4. Energy level 1
5. Energy level 3
6. No because the valence shell is full
7. 1, 0, no because an electrons was removed and there are more protons
8. hydrogen, reactive remove, accept or share
neon non reactive, remain the same
carbon reactive, share
fluorine, reactive, accept
sodium, reactive, lose
2. No because balanced electrons won't gain or lose electrons
3. They have 4 valence electrons
4. Energy level 1
5. Energy level 3
6. No because the valence shell is full
7. 1, 0, no because an electrons was removed and there are more protons
8. hydrogen, reactive remove, accept or share
neon non reactive, remain the same
carbon reactive, share
fluorine, reactive, accept
sodium, reactive, lose